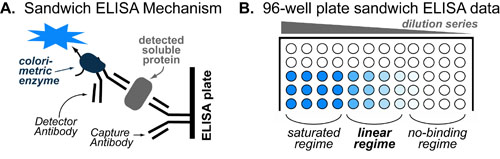
ELISA or Enzyme-Linked Immunosorbent assay is the most commonly used method for measuring proteins concentrations in solution. It is extensively used both in the laboratory (e.g. culture supernatant) and the clinic (e.g. blood tests) due to its simplicity and adaptability to other protein-based assays such as high throughput screening. The way it works is outlined in the figure above and described in detail below:
- Capture Antibody:Coat a 96-well plate overnight with your “capture antibody” (which captures your protein of interest)
- Block plate: Wash plate and block plate with BSA (bovine serum albumin) solution for 1 hour (this protein reduces the stickiness of parts of your 96-well plate that aren’t coated with capture antibody)
- Dilution series: Wash plate and add dilution series of your biological fluid for 1 hour (it is necessary to run a dilution series and not a single concentrations for reasons discussed below)
- Detector Antibody:Wash plate and add ~1/1000 dilution of your “detector antibody” for 1 hour (this antibody has an enzyme attached to it which will produce color that is proportional to the amount of protein you with to measure)
- Add substrateWash plate and add substrate for your “colorimetric enzyme” that is attached to your detector antibody
- Stop reaction: When your plate looks like that figure above, stop the colorimetric reaction with acid.
- Read plate: Exactly measure the amount of color per well using a plate reader
In general, the most important part of your plate/data is the “linear regime” of the dilution series. This regime is where the amount of color is proportional to the concentration of protein (see post on the Hill Equation). The color/absorbance can be converted to an exact concentration using a calibration curve (with standard/pure protein) which must be on the same plate.
Aside from measuring soluble protein, ELISA can also be used to measure protein-protein binding constants using the Hill Equation and the Cheng-Prusoff Equation. While ELISA binding curves are usually sigmoidal (or S-shaped) as that theory requires, they sometimes exhibit bell-like shapes. This effect, is known as the “prozone phenomenon” or “hook effect” and is a result of the fact: ELISA relies on the formation of a three component (ternary) complex between the (1) capture antibody (2) protein of interest and (3) detector antibody. In these cases the equation below must be used to fit the curves and obtain a binding constant:
For more information on ternary complexes you can read my first paper from graduate school exactly characterized ternary complex equilibria (solving a problem in theoretical biochemistry that had previously been described as mathematically “unsolvable”).
REFERENCES:
- Howard, G.C.; Kaser, M.R. Making and Using Antibodies: A Practical Handbook CRC Press 2006
- Alberts, B. Molecular Biology of the Cell 5th Ed. Garland Science 2008
- Murphy, K. Janeway’s Immunobiology 8th Ed. Garland Science 2012
- Taborda, C.; Rivera, J.; Zaragoza, O.; Casadevall, A. More Is Not Necessarily Better: Prozone-Like Effects in Passive Immunization with IgG. J. Immunol. 2003, 170, 3621-3630.
- Braun, A.; Alsenz, J. Development and use of enzyme-linked immunosorbent assays (ELISA) for the detection of protein aggregates in interferon-alpha (IFN-α) formulations. Pharm. Res. 1997, 14, 1394
- Rodbard, D.; Feldman, Y.; Jaffe, M. L.; Miles, L. E. M.; Kinetics of two-site immunoradiometric (sandwich) assays II: Studies on the nature of the-high-dose hook effect. Immunochem. 1978, 15, 77-82
- Douglass Jr., E.F.; Miller, C.J.; Sparer, G.; Shapiro, H.; Spiegel, D.A.; A Comprehensive Mathematical Model for Three-Body Binding Equilibria. J. Am. Chem. Soc. 2013, 135, 6092-6099.
- Andersen, P.S.; Schuck, P.; Sundberg, E.J.; Geisler, C.; Karjalainen, K.; Mariuzza, R.A. Quantifying the Energetics of Cooperativity in a Ternary Protein Complex. Biochemistry, 2002, 41, 5177-5184.
- Otsuki, J.; Narita, T.; Tsutsumida, K.; Takatsuki, M.; Kaneko, M. Modular Approach toward Supramolecular Functional Assemblies: Characterization of Donor− Spacer− Acceptor Ternary Complexes. J. Phys. Chem. A. 2005, 109, 6128-6134.
- Streusand, V.; Björk, I; Gettins, P; Petitou, M; Olson, S.T. Mechanism of acceleration of antithrombin-proteinase reactions by low affinity heparin. J. Biol. Chem. 1995, 270, 9043-9051.

This work by Eugene Douglass and Chad Miller is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.