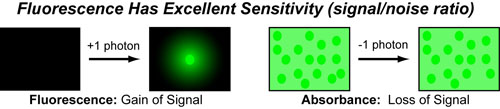
In a follow-up to our introduction to fluorescence, we wanted to discuss why fluorescent probes have proven so useful in chemistry and biology. One of the main reasons is that, unlike most spectroscopic techniques which rely on a loss-of-signal or light-absorption, fluorescence is a “gain of signal” technique. As a result, the near-zero baseline/background translates into a very high signal to noise ratio for fluorescent probes. Indeed fluorescent probes have some of the greatest sensitivities of all sensors (radiation is better but has other draw backs)!
In general, the basic principle behind fluorescent sensors is: (1) the phenomenon you are studying causes a change in the molecular structure/environment/etc. of the fluorophoe and (2) this molecular change causes a change in magnitude or shape of the fluorophores spectrum. These changes can be calibrated to give you exact quantification of your phenomena of interest.
REFERENCES:
- Lakowicz, J. R. in Principles of Fluorescence Spectroscopy, Springer, 2006
- McQuarrie, D. A.; Simon, J. D. Physical Chemistry: A Molecular Approach., University Science Books, 1999
- Skoog, D. A., Holler, F. J. & Nieman, T. A. Principles of Instrumental Analysis (5th ed), Saunders College Publishing, 1998