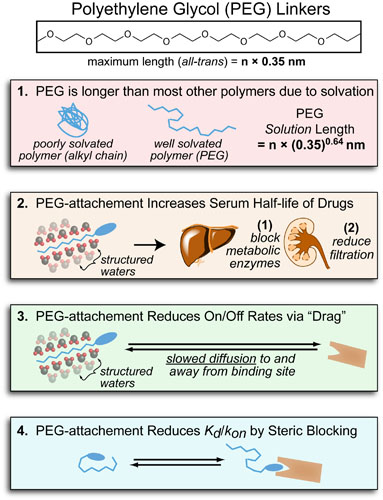
Where synthetic chemistry has given us many molecules that bind (and inhibit) many different proteins, chemical biology endeavors to “attach” new function to these “classical” drugs. Examples of chemical biological applications include: (1) attaching toxins or imaging agents for targeted deliver or (2) using multivalency to improve a drug’s potency. Unfortunately in order to “attach” new function to a drug you need to use a “linker” which is long and inert so it doesn’t interfer with “binding” or the new “function”. The most common linker material used in chemical biology and pharmacology is polyethylene glycol or PEG (pictured above) which is both long and inert but still impacts the properties of the drugs it is attached to(see figure above):
- PEG is longer than other polymers: Though PEG is n x 0.35nm long in its all-trans conformation (where n is the number of ethylene glycol units) its average solution length is n x (0.35)0.64nm. While this length is shorter it is still longer than most other linker materials which tend to “ball up” in aqueous solution due to the fact they are more hydrophobic than PEG (i.e. less well solvated than PEG).
- PEG increases how long drugs stay in the body: An additional advantage of PEG’s high solvation is that it helps prevent the liver and kidneys from removing an attached drug from the body. This is done by (1) sterically blocking metabolic enzymes in the liver from interacting with (and destroying) the drug and (2) increasing the effective size of the drug so the kidneys can’t filter it out (into the urine).
- PEG slows a drug’s diffusion: One disadvantage of PEG carrying all this “structured water” is that it slows down the drugs ability to “explore” its environment (diffuse). In general, this “diffusion effect” reduces both the kon and koff of a drug equally and has no effect on its potency (EC50) or Kd. Practically, however, this could mean you need to increase the incubation times in your experiments.
- PEG reduces a drugs potency by blocking binding: Though diffusion effects have no effect on a drugs EC50 or Kd, sufficiently long PEG-linkers can block binding of the drug to its target by “wrapping around” the drug. This reduces the kon and makes the EC50 of the drug worse.
REFERENCES:
- Baird, E.; Holowka, D.; Coates, G.; Baird, B. Highly effective poly (ethylene glycol) architectures for specific inhibition of immune receptor activation. Biochem. 2003, 42, 12739-12748
- Glue, P. Pegylated interferon-α2b: Pharmacokinetics, pharmacodynamics, safety, and preliminary efficacy data. Clin. Pharm. Therap. 2000, 68, 556-567
- Kubetzko, S.; Sarkar, C.; Plückthun, A. Protein PEGylation decreases observed target association rates via a dual blocking mechanism. Mol. Pharm., 2005 68, 1439
- Dhalluin, C. et al. Structural and biophysical characterization of the 40 kDa PEG-interferon-α2a and its individual positional isomers. Bioconj. Chem., 2005, 16, 504-517
- Pepinsky, R. et al. Design, synthesis, and analysis of a polyethelene glycol-modified (PEGylated) small molecule inhibitor of integrin α4β1 with improved pharmaceutical properties. J. Pharm. Exper. Therap., 2005, 312, 742
- Das, R. et al. Binding mechanisms of PEGylated ligands reveal multiple effects of the PEG scaffold. Biochem. 2008, 47, 1017-1030
- Lu, Y. et al. Effect of PEGylation on the solution conformation of antibody fragments. J. Pharm. Sci. 2008, 97, 2062-2079
- Kang, J.; DeLuca, P.; Lee, K. Emerging PEGylated drugs. Expert Opin Emerg Drugs. 2009 14, 363-80
- Jevševar, S.; Kunstelj, M. PEGylation of therapeutic proteins. Biotech. J. 2010, 5, 113-128
- Pai, S. et al. The Conformation of the Poly (ethylene glycol) Chain in Mono-PEGylated Lysozyme and Mono-PEGylated Human Growth Hormone. Bioconj. Chem. 2011, 16, 2317-2323
- Jeppesen, C. et al. Impact of polymer tether length on multiple ligand-receptor bond formation. Science, 2001, 293, 465
- Rudnick, J.; Gaspari, G. The shapes of random walks. Science, 1987, 237, 384
- Oesterhelt, F.; Rief, M.; Gaub, H. Single molecule force spectroscopy by AFM indicates helical structure of poly (ethylene-glycol) in water. New J. Phys. 1999, 1, 6

This work by Eugene Douglass and Chad Miller is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.