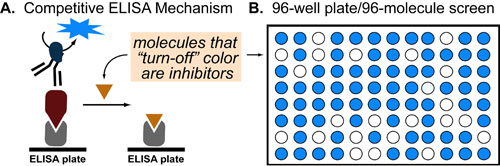
In a follow-up to our post introducing ELISA, we wanted to discuss a common application of this technique: small molecule inhibitor screening. The set up is relatively simple (1) Coat a 96-well plate with your two proteins of interest and and a detector antibody (2) add a library of 96 molecules per plate (3) Inspect which molecules inhibit the protein-protein interaction (resulting in a color loss).
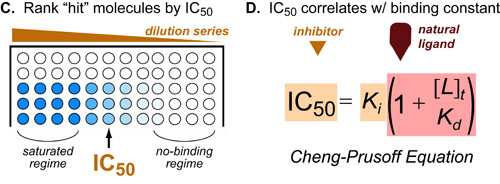
Once you have have narrowed down your library to a list of inhibitors, you can rank those inhibitors by their potency/IC50 by running dilution series with the exact same plate set-up:
These IC50’s directly correlate with the binding constants (Ki) of your inhibitors. See our post on competitive binding experiments for more information on these type of experiments.
REFERENCES:
- Cheng, Y.; Prusoff, W.H. Relationship between the inhibition constant (KI) and the concentration of inhibitor which causes 50% inhibition (IC50) of an enzymatic reaction. Biochem. Pharmacol. 1973, 22, 3099-3108.
- Howard, G.C.; Kaser, M.R. Making and Using Antibodies: A Practical Handbook CRC Press 2006
- Lauffenburger, D.A. Receptors: Models for Binding, Trafficking and Signalling, Oxford University Press 1993.
- Segel, I.H. Enzyme Kinetics: Behavior and Analysis of Rapid Equilibrium and Steady-State Enzyme Systems, Wiley-Interscience, 1993
- Alberts, B. Molecular Biology of the Cell 5th Ed. Garland Science 2008
- Murphy, K. Janeway’s Immunobiology 8th Ed. Garland Science 2012

This work by Eugene Douglass and Chad Miller is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License.